Do you have Alport Syndrome?
A new Australian clinical trial might be right for you.
MONASH HEALTH HOSPITAL – ELOXX CLINICAL TRIAL
Monash Health Hospital is looking for small group of Alport patients aged 6-30 with certain gene changes (nonsense mutation) to participant in a clinical trial of testing a new treatment
Eloxx company is testing a new treatment that they think might help certain people with Alport Syndrome. Alport syndrome is caused by changes (called mutations) in certain genes. Only a very small proportion of gene changes (called ‘nonsense mutations’ which account for approximately 3-5% that cause Alport syndrome) are potentially treatable by this new testing treatment (called ELX-02). The main goal of this study (called EL-014) is to learn whether ELX-02 is safe in Alport patients and whether it improves kidney function and hearing. The study will soon open trial sites in the UK and Australia.
Please note that the study team will need to know your genetic test result and latest kidney function reading (called eGFR) to be able to tell whether you might be eligible for this study.
Details of sites running the Eloxx clinical trial:
Australia – Monash Health, Melbourne (adults) – Prof. Kerr – Clinresneph@monashhealth.org.
Note: Criteria for people appropriate for the Eloxx study.
The study will be performed in Australia and UK and is looking for male and female participants who:
-
• Are 6-30 years of age (inclusive)
• Have a confirmed diagnosis of X-linked Alport syndrome with a nonsense mutation in COL4A5, or a diagnosis of autosomal recessive Alport syndrome with two nonsense mutations in either COL4A3 or COL4A4
• Are on a stable dose of an ACE inhibitor or ARB for 4 weeks or more
• Have over a certain value of kidney filtration rate (eGFR >60ml/min/1.73m2)
• Have not received a kidney transplant
• Live in UK and Australia or willing to travel to those countries
• Available for 60 days treatment with bi-weekly visits, followed by 3 months with monthly visits
HERA CLINICAL TRIAL
Are you or someone you know living with Alport Syndrome? If you have been diagnosed with Alport Syndrome, you may be eligible to participate in a clinical trial investigating a new drug for adult patients with Alport Syndrome.
The clinical trial will look to see how well a new experimental drug works in preserving kidney function. This treatment is only available to patients participating in the clinical trial.
You may be eligible for this trial if:
-
- You are aged 18-55 years old
- You have confirmed Alport Syndrome via genetic testing
- You have kidney disease
- You do not have diabetes
- You are less than 110kg
TO LEARN MORE PLEASE CONTACT:
Donna North: Royal Melbourne Hospital, Victoria, Australia.
email: donna.north@mh.org.au
Belinda Elford: Royal Brisbane & Womens Hospital, Queensland, Australia.
email: belinda.elford@health.qld.gov.au
Aron Chakera. Sir Charles Gairdner Hospital, WA, Australia.
Phone: aron.chakera@health.wa.gov.au

REATA GLOBAL PHASE 2/3 TRIAL IN CHRONIC KIDNEY DISEASE CAUSED BY ALPORT SYNDROME
Reata believes bardoxolone methyl has the potential to address the underlying causes of GFR loss in Alport syndrome patients because it activates molecular pathways that promote the resolution of inflammation by restoring mitochondrial function, reducing oxidative stress, and inhibiting ROS-mediated pro-inflammatory signaling. These anti-inflammatory and tissue-protective effects suppress multiple cellular processes that conspire to promote GFR loss. Bardoxolone methyl and closely related structural analogs have been shown to improve renal function, reduce inflammation, and prevent injury, remodeling, and fibrosis in many animal models of renal injury and disease.
Bardoxolone methyl has been tested in seven studies of patients with CKD caused by type 2 diabetes that enrolled approximately 2,600 patients. These studies included a randomized, placebo-controlled 52-week Phase 2b study (“BEAM”) and a large, multinational Phase 3 study (“BEACON”) that enrolled only patients with severe (Stage 4) CKD. In these studies, bardoxolone methyl treatment significantly increased eGFR and creatinine clearance, significantly reduced uremic solutes (BUN, uric acid, and phosphate) in inverse correlation to eGFR increases, and numerically reduced renal SAEs and ESRD events.
The ATHENA Study: Mapping the Course of Alport Syndrome
The ATHENA study was designed to learn more about the progression of Alport syndrome, particularly regarding the changes in the kidneys over time in Alport syndrome patients. The ATHENA study was an observational study and did not involve the use of any investigational drugs. However, information obtained from this study will help in the design of future clinical trials to test a new investigational drug in patients with Alport syndrome.
Patients with chronic kidney disease due to Alport syndrome, particularly women and those with the X-linked form of the disorder, have a low quality of life that decreases over time, according to a natural history of disease study. Findings of the study, conducted by Regulus Therapeutics, also revealed new urine and blood biomarkers correlating with the decline of kidney function in these patients.
Regulus presented two posters on its international, multicenter, observational study (NCT02136862), called ATHENA, at the American Society of Nephrology’s recent Kidney Week 2018 meeting in San Diego. A total of 165 patients with a mean age of 44.8 years were included, of whom 33.9% were men, 64.8% had X-linked Alport, and 83% were white. The patients were followed up for two years. Read more…

Research Funded by the Alport Foundation of Australia
Using induced pluripotent stem cell-derived podocytes to create an in vitro model of Alport Syndrome
Monash University/Dr John Haynes & Prof Sharon Ricardo
A significant feature of Alport Syndrome is the death of podocytes, the cells that help regulate filtration in the kidney. The widely accepted view is that, due to a mutation in one of the genes responsible for the manufacture of cellular collagen, inappropriately folded collagen accumulates inside the cell, placing it under stress. Eventually the protective mechanisms employed by the podocytes to cope with the stress are overwhelmed and the cells die. The body finds it difficult to replace lost podocytes, this forces the surviving podocytes to work harder, placing them under even more stress.
We are looking at this process in a slightly different way. The collagens produced by the cells not only help secure the cells to surfaces, they also signal within cells in their own right. Our contention is that disruptions of normal cellular signalling place podocytes under more stress than previously considered. This project investigates the contribution of collagen signalling to cell survival in healthy podocytes and in podocytes derived from individuals with Alport Syndrome.
Kidneys in a dish: Scientists reprogram adult skin cells to make mini kidneys
Posted 8 Oct 2015
Adult skin cells have been reprogrammed to make the most mature human kidneys yet to be grown in a dish, say researchers.
The mini kidneys have hundreds of filtering units and blood vessels and appear to be developing just as kidneys would in an embryo.
“The short-term goal is to actually use this method to make little replicas of the developing kidney and use that to test whether drugs are toxic to the kidney,” said lead researcher Professor Melissa Little, of the Murdoch Childrens Research Institute.
“Ultimately we hope we might be able to scale this up so we can … maybe bioengineer an entire organ.”
Read more… http://www.abc.net.au/news/2015-10-08/proto-kidney-grown-in-a-dish/6833882
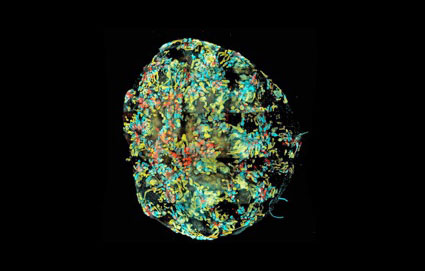
Making stem cell lines from patients with Alport Syndrome
Prof Sharon Ricardo, Monash University (2013)
The epidemic of chronic kidney disease and end-stage renal failure represents a crisis for health world-wide. Alport syndrome is an inherited kidney disease that accounts for 2% of all patients with end-stage renal failure. Most mutations in the common X-linked form, result in missense or nonsense changes, but little is known of how these cause disease or how it might be modified.
Currently, there is no satisfactory model in which to examine the effect of mutations in X-linked Alport syndrome or to evaluate new treatments. Furthermore, human kidney cells called podocytes, the main cells involved in blood filtration, are very difficult to establish due to their limited ability to replicate and maintain in culture.
However this has recently changed. The reprogramming of adult cells to generate induced pluripotent stem cells (iPS), are valuable for use in disease modelling, drug screening and regenerative medicine – as recently reviewed (O’Neill and Ricardo JASN 2013). Funding from the Alport Foundation of Australia is supporting the generation of iPS cell lines generated by obtaining cells, whether skin or urine, from patients with Alport Syndrome. We will use iPS cells that are genetically tailored to patients with Alport Syndrome to assess how missense and nonsense COL4A5 mutations in X-linked Alport syndrome produce disease. Understanding these mechanisms is the first step in developing new disease-modifying treatments.

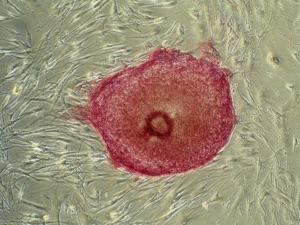
Novel stem cell assays for Alport Syndrome
Project research grant awarded to CIs Ricardo and Kerr by the Alport Foundation of Australia, May 2010.
The potential of stem cells represents one of the greatest opportunities in medicine. They are found in embryonic tissues and adult organs and have potential uses in therapies designed to repair and regenerate organs. Recently, scientists have discovered how to reprogram adult cells by introducing four regulatory genes causing them to lose their characteristic adult properties and behave more like embryonic stem cells. These cells termed inducible pluripotent stem cells, or iPS cells, become more immature in nature and can be maintained in a lab dish indefinitely. The establishment of iPS cell lines from patients with Alport Syndrome has an extraordinary potential for new drug discovery and personalized medicine. This is where the study of iPS cells lines could provide information about an individual patient to select or optimize that patient’s preventative and therapeutic care. It will also enable us to understand Alport Syndrome in a way we’ve never been able to before, advancing the potential of human iPS cells for modelling the genetic disorder and for the screening of new drug compounds. Read more about Dr Ricardo…..
Novel Chemical Treatments for Alport Syndrome
Project research grant awarded to J Savige by the Alport Foundation of Australia, May 2010.
Alport syndrome is an inherited disease that results in progressive kidney failure, hearing loss and eye abnormalities. It is due to mutations in 3 genes that code for different collagen chains. These chains are normally intertwined to form a single molecule in basement membranes in the kidney, ear and eye. The different forms of Alport syndrome affect different chains. Each type results in the destruction not only of the corresponding mutant chain but also of the chains with which it is normally intertwined. Thus in Alport syndrome all 3 chains are missing from affected basement membranes. These membranes are consequently abnormal and weakened and the abnormal membranes result in the clinical features of kidney disease, hearing loss and ocular abnormalities.
We know that some chemicals potentially inhibit the breakdown of the defective collagen chains and we hypothesise persistence of even partly abnormal chains in the basement membranes will result in less severe disease. We have preliminary evidence that this is so. Here we seek funding to look at cell lines from patients with.

Different kinds of mutations causing X-linked and autosomal recessive Alport syndrome to show these chemicals actually increase the amount of the collagen mRNA and protein. We will compare several chemicals and determine which is most effective in doing this. This approach is already being trialled in clinical studies in cystic fibrosis.
The only realistic treatments available for Alport syndrome currently focus on minimising proteinuria and delaying the onset of end-stage renal failure. The most commonly-used agents are angiotensin converting enzyme (ACE) inhibitors. The use of chemical chaperones and inhibitors of nonsense-mediate decay represents a new approach to the treatment of Alport syndrome. There are early clinical trials of some of these agents in cystic fibrosis and there is also hope they will be useful in neurodegenerative diseases (Chaudhuri 2006). The proposed project will provide proof of principle that chemical chaperones and inhibitors of nonsense mediated decay increase the amount of functional collagen IV a5 chain.


